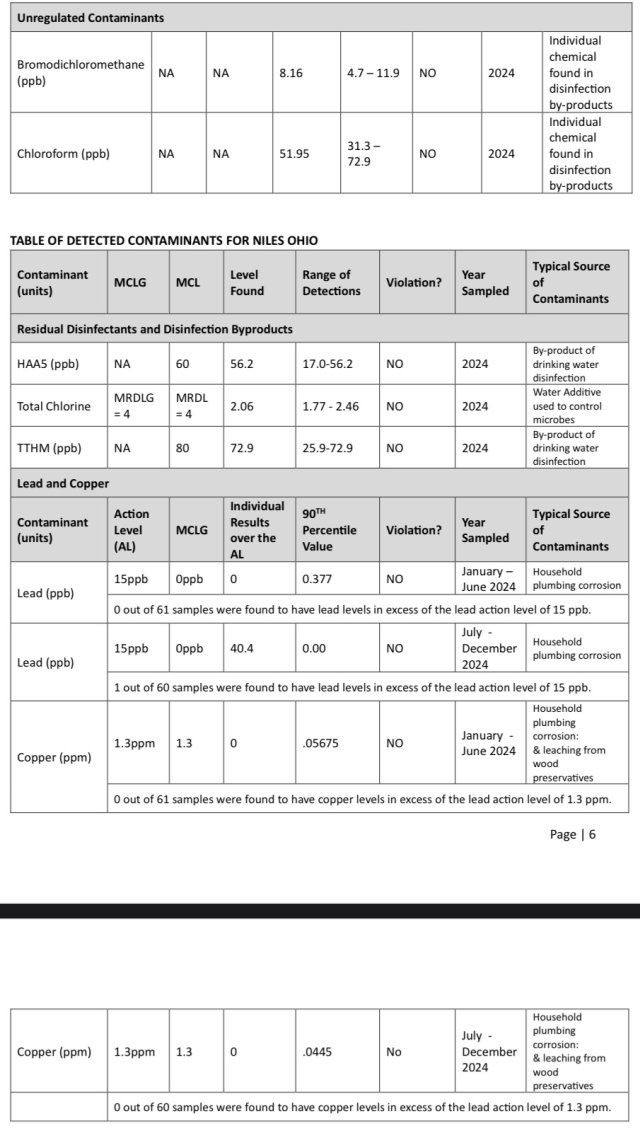
Before u installed the injector pump what were your findings while dripping "tainted" water lol? Anything noticeably detrimental or did u just add it in for more of worry/precaution?Howdy
When i used municipal water i had an injector pump on my drip, which automatically dosed Prime. Its flow-driven, no concerns during power outage. No need for an offgassing reservoir, either, just a bucket or a carboy with diluted prime
Works virtually maintenance free.

Chemilizer™ HN55 Fixed Ratio Injectors
The Chemilizer HN55 proportional liquid injector runs off of water flow, rather than electricity. Use for disinfectants, fertilizers, biologicals and more.www.qcsupply.com
HarleyK
Chloramine in source water with Auto Drip System?
- Thread starter wednesday13
- Start date
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
IMO chlorine poisoning is less of an issue with an establish filter compared to the immediate danger your fish will be in. You'd be hard pressed to find any natural environment, outside of physical extremes, where micro-biological life cannot exist and this is reflected even with the bacterial colonies in our home aquariums.Is it possible in your opinion for a filter to be so dirty everything stays stable and "cycles" out even if some of the bacteria is dying? I don't clean anything for years and years and ive always found if i clean too much or drip too much fresh water a system can crash super quickly. I'm honestly starting to think ive been running with chloramines for several years if not the whole time and not even noticing. Or noticing in my own way indirectly. Too much fresh water equates to higher nitrates and that would mean in my eyes my filters/systems cant break down that much new ammonia quickly enough so turn the drip down. I started by cutting my drips all together and nitrates dropped in the first 24hrs. I let that ride for almost 2 weeks before they went up again. After that i returned to dripping at 100-150% a week over time to restore nitrates to 10-20 and back to "normal". Trying to use as little water as possible now to add less chlorine/chloramine but still keep nitrates under 20. My tanks are 230,450,500,650... as i mentioned in another comment im "testing" with a couple 40 breeders at 200% weekly tank volume replacement trying to create a problembut tests are all still good.
Most bacteria colonies in an establish filter will have a natural bio-film (think of it as a physical barrier) that will protect against the immediate washing of even untreated tap water. Of course if you repeatedly scrub it down or soak the bio-media for an extended period you could probably kill 99.9% of your colony but a quick rinse most likely won't kill everything instantly. Again, this has to do with the concentration of the solution as I mentioned in a previous post; chlorine will only kill a cell if it can access/enter the cell membrane so it's a game of probability. Of course this statement doesn't tell the full truth but for the purpose of explaining to the layman it is good enough.
The issue with a fish compared to bacteria is that the gills have evolved to become an interdependent organ to a larger organism; it's specialized in a specific way to assist in keeping the larger organism alive rather than itself. The hope being the organism as a whole will have other specialized cells/organs who can assist it keeping the other cells/organs alive. On the flip side, bacteria are single-cellular self-reproducing organisms so their defenses maximize for the individual which coincidentally helps keep the colony alive.
You can think of it like this: an organ is like a bus, a bacteria colony is like a car, and people are individual cells. You can fit a lot of people inside a bus but you can field more cars then busses in a given amount of space. Both vehicles are protected against glancing outside threats due to their metal exteriors but obviously overwhelming force could bypass this defense. However, what if we say 50% of the cars have their windows open and 50% of the windows on the bus are open. If a thick chlorine gas cloud suddenly appeared and spread through this population of cars and 1 bus it is clear that at most only 50% of the cars will be affected but the bus will definitely have all of its people affected. Ergo, 50% of the cars will remain fine, the other 50% are injured or dead, and the entire bus is now in mayhem as there is a mix of people dying and severely injured. In other words, leaving the analogy, while half of the bacteria colonies may end up dead at least half survived compared to the organ which has severe, possible permanent damage.
Of course in that example I used an extremely high concentration of chlorine gas with guaranteed contact to make a point but the real expanation isn't as simple and clean as the hypothetical above. The main points still stand though, at a high enough concentration you may not see all your bacteria colonies die off but your fishes' gills will definitely be affected.
To help people understand further, if they want, below is where I will describe why "chlorine poisoning" occurs and how it leads to cellular death. Although it won't be a 100% in-depth explanation I hope it will explain the chemical & physical reactions occurring well enough for everyone.
Chlorine (CL) is a naturally occurring highly reactive (meaning it wants to bond) chemical element; it is actually a vital building block of life for most plants and animals on this planet. Of course in nature due to it being so reactive you will almost never find it naturally in it's mono-elemental state but rather mixed with something else like sodium chloride (table salt). As a result when CL is mixed with water to be a disinfectant they use chlorine gas (CL2). When CL2 mixes with water we get mostly hypochlorous acid (HOCL) and hydrochloric acid (HCL) with some leftover undissolved CL2. Typically the leftover CL2 will gas off at the earliest opportunity due to the water and air trying to reach equilibrium but the other two chemicals typically stay to some degree.
First, HCL is extremely water-soluble so it immediately turns into hydronium (H3O+ but you will often see H+ as shorthand in scientific literature) and chloride (CL-) ions when introduced to water. While this reaction does generate quite a bit of heat and is acidic, -> lower pH, the amount created from CL2 is so miniscule in normal home aquarium setups this part of the reaction is just here for a more complete explanation. Unless you directly pump CL2 into you tank you should never have to worry about HCL.
Next, HOCL is partially water-soluble so it can eventually break into separate chemicals producing H+ and hypochlorite ions (OCL-). However, this reaction is not instantaneous and is pH dependent ( ph <7.5 more HOCL stays; pH >7.5 more OCL- formed). Both HOCL and OCL- are considered disinfectants but due to their chemical composition they behave differently when interacting with a cell membrane or cell wall.
To understand the next explanation first you need to know that most cell membranes are amphipathic meaning they have hydrophillic ("water-loving") and hydrophobic ("water-fearing") parts to them. Typically the hydrophillic part of the membrane is on the outside while the hydrophobic parts are on the inside. In addition almost all cell membranes will have a negative charge associated with them on the outer layer and all of this is fairly compact. As a result, only relatively small neutral or positively charged molecules can typically enter a cell membrane.
Now, since HOCL has a neutral charge most cell membranes do not push it away when it comes close enough for contact. This results in the molecule slipping past the cell membrane which inevitably causes an oxidation reaction with the DNA, proteins, or essential enzymes causing the cell to die but also results in the HOCL molecule to decompose into non-reactive forms. OCL- could perform in a similar manner but due to it's negative charge it attracts water molecules to it forming a "shell" of sorts. As a result OCL- ions are both negatively charged (and therefore pushed away due to the cell membrane's charge) and too large to easily enter a cell.
Final note, monochloramine, the chloramine most commonly used to treat water, can also form in your home aquarium if HOCL and NH3 (ammonia) (or CL2 and NH4+ [ammonium]) is present. Therefore, it's always best practice to dechlorinate your water with chemicals before adding it to your tank. Unfortunately if you do not use a dechlorinator, even if your tank has 0 ammonia available, a small amount of the HOCL will turn back to CL2 and gas off but most will likely turn into OCL-. This chemical will not degrade unless it reacts with left over organics or unless UV light is hitting it turning it into O(xygen)2 and CL-. If you really don't want to use any chemicals at all for dechlorination and you know for a fact you only have chlorine in your water then a wave maker/surface fan + open bucket/container +UV light is all required over the course of several days to ensure 100% dechlorination has occured. OCL- is less reactive than HOCL so if you're fine with good enough dechlorination with no chemicals, 24-48 hours is also enough.
The question is then why do fish die from "chlorine poisoning", there isn't enough chlorine in 2-4ppm to cause total body cellular death right? And that is 100% true. The issue of "chlorine poisoning" is the servere and potentially permanent scarring of the gills for our fish (with an extremely low chance of direct internal organ & blood vessel damage). Since a fish's gills act as lungs they quickly sufficate if the damage is too servere.
Let me know if you have anymore questions.
HTH
Last edited:
IMO chlorine poisoning is less of an issue with an establish filter compared to the immediate danger your fish will be in. You'd be hard pressed to find any natural environment, outside of physical extremes, where micro-biological life cannot exist and this is reflected even with the bacterial colonies in our home aquariums.
Most bacteria colonies in an establish filter will have a natural bio-film (think of it as a physical barrier) that will protect against the immediate washing of even untreated tap water. Of course if you repeatedly scrub it down or soak the bio-media for an extended period you could probably kill 99.9% of your colony but a quick rinse most likely won't kill everything instantly. Again, this has to do with the concentration of the solution as I mentioned in a previous post; chlorine will only kill a cell if it can access/enter the cell membrane so it's a game of probability. Of course this statement doesn't tell the full truth but for the purpose of explaining to the layman it is good enough.
The issue with a fish compared to bacteria is that the gills have evolved to become an interdependent organ to a larger organism; it's specialized in a specific way to assist in keeping the larger organism alive rather than itself. The hope being the organism as a whole will have other specialized cells/organs who can assist it keeping the other cells/organs alive. On the flip side, bacteria are single-cellular self-reproducing organisms so their defenses maximize for the individual which coincidentally helps keep the colony alive.
You can think of it like this: an organ is like a bus, a bacteria colony is like a car, and people are individual cells. You can fit a lot of people inside a bus but you can field more cars then busses in a given amount of space. Both vehicles are protected against glancing outside threats due to their metal exteriors but obviously overwhelming force could bypass this defense. However, what if we say 50% of the cars have their windows open and 50% of the windows on the bus are open. If a thick chlorine gas cloud suddenly appeared and spread through this population of cars and 1 bus it is clear that at most only 50% of the cars will be affected but the bus will definitely have all of its people affected. Ergo, 50% of the cars will remain fine, the other 50% are injured or dead, and the entire bus is now in mayhem as there is a mix of people dying and severely injured. In other words, leaving the analogy, while half of the bacteria colonies may end up dead at least half survived compared to the organ which has severe, possible permanent damage.
Of course in that example I used an extremely high concentration of chlorine gas with guaranteed contact to make a point but the real expanation isn't as simple and clean as the hypothetical above. The main points still stand though, at a high enough concentration you may not see all your bacteria colonies die off but your fishes' gills will definitely be affected.
To help people understand further, if they want, below is where I will describe why "chlorine poisoning" occurs and how it leads to cellular death. Although it won't be a 100% in-depth explanation I hope it will explain the chemical & physical reactions occurring well enough for everyone.
Chlorine (CL) is a naturally occurring highly reactive (meaning it wants to bond) chemical element; it is actually a vital building block of life for most plants and animals on this planet. Of course in nature due to it being so reactive you will almost never find it naturally in it's mono-elemental state but rather mixed with something else like sodium chloride (table salt). As a result when CL is mixed with water to be a disinfectant they use chlorine gas (CL2). When CL2 mixes with water we get mostly hypochlorous acid (HOCL) and hydrochloric acid (HCL) with some leftover undissolved CL2. Typically the leftover CL2 will gas off at the earliest opportunity due to the water and air trying to reach equilibrium but the other two chemicals typically stay to some degree.
First, HCL is extremely water-soluble so it immediately turns into hydronium (H3O+ but you will often see H+ as shorthand in scientific literature) and chloride (CL-) ions when introduced to water. While this reaction does generate quite a bit of heat and is acidic, -> lower pH, the amount created from CL2 is so miniscule in normal home aquarium setups this part of the reaction is just here for a more complete explanation. Unless you directly pump CL2 into you tank you should never have to worry about HCL.
Next, HOCL is partially water-soluble so it can eventually break into separate chemicals producing H+ and hypochlorite ions (OCL-). However, this reaction is not instantaneous and is pH dependent ( ph <7.5 more HOCL stays; pH >7.5 more OCL- formed). Both HOCL and OCL- are considered disinfectants but due to their chemical composition they behave differently when interacting with a cell membrane or cell wall.
To understand the next explanation first you need to know that most cell membranes are amphipathic meaning they have hydrophillic ("water-loving") and hydrophobic ("water-fearing") parts to them. Typically the hydrophillic part of the membrane is on the outside while the hydrophobic parts are on the inside. In addition almost all cell membranes will have a negative charge associated with them on the outer layer and all of this is fairly compact. As a result, only relatively small neutral or positively charged molecules can typically enter a cell membrane.
Now, since HOCL has a neutral charge most cell membranes do not push it away when it comes close enough for contact. This results in the molecule slipping past the cell membrane which inevitably causes an oxidation reaction with the DNA, proteins, or essential enzymes causing the cell to die but also results in the HOCL molecule to decompose into non-reactive forms. OCL- could perform in a similar manner but due to it's negative charge it attracts water molecules to it forming a "shell" of sorts. As a result OCL- ions are both negatively charged (and therefore pushed away due to the cell membrane's charge) and too large to easily enter a cell.
Final note, monochloramine, the chloramine most commonly used to treat water, can also form in your home aquarium if HOCL and NH3 (ammonia) (or CL2 and NH4+ [ammonium]) is present. Therefore, it's always best practice to dechlorinate your water with chemicals before adding it to your tank. Unfortunately if you do not use a dechlorinator, even if your tank has 0 ammonia available, a small amount of the HOCL will turn back to CL2 and gas off but most will likely turn into OCL-. This chemical will not degrade unless it reacts with left over organics or unless UV light is hitting it turning it into O(xygen)2 and CL-. If you really don't want to use any chemicals at all for dechlorination and you know for a fact you only have chlorine in your water then a wave maker/surface fan + open bucket/container +UV light is all required over the course of several days to ensure 100% dechlorination has occured. OCL- is less reactive than HOCL so if you're fine with good enough dechlorination with no chemicals, 24-48 hours is also enough.
The question is then why do fish die from "chlorine poisoning", there isn't enough chlorine in 2-4ppm to cause total body cellular death right? And that is 100% true. The issue of "chlorine poisoning" is the servere and potentially permanent scarring of the gills for our fish (with an extremely low chance of direct internal organ & blood vessel damage). Since a fish's gills act as lungs they quickly sufficate if the damage is too servere.
Let me know if you have anymore questions.
HTH

Appreciate the time ur taking to help me out
Am I misunderstanding that chlorine/chloramine poisoning isnt a quick process? IMO keeping anything alive over 10yrs is a feat in itself with regular things like power outtages and equipment failure and i have plenty heads here over the decade mark living in what is most likely chlorine/chloramine source water being doused right on them 24/7.
Ultimately, what is the exact kind of test i need to be doing in my tanks to see if im dealing with chlorine or chloramine and if its even worth mitigating.
Im just not seeing anything like loss of appetite or stunted growth, cloudy eyes, labored breathing etc…
Chlorine/chloramine poisoning on the cellular level is a near instant process if the molecule gets through the cell wall but the threshold for a death sentence is going to depend on what species of fish you have, the concentration, and how long the fish has been exposed to the toxin. Unfortunately this mean a range of 0.05-0.5 ppm chlorine could cause severe/permanent damage on a scale of most sensative -> least sensative but the timeline from poisoning to death could be as fast as a few minutes in the most extreme case to a week.View attachment 1573346
Appreciate the time ur taking to help me out… Def. understand worst case scenario. Just trying to figure out if my source is bad enough to need chemical improvement. Not opposed to chemicals either if it comes to that. If things can be as bad as ur saying i feel like i should have alot of dead fish on my hands already. 95% of the fish here are ancients and breathe air so technically any gill damage wouldnt matter as much lol (joking)… i cant just be that lucky tho to think i havent been dealing with this for years or at the very least over a year since ive experienced more water main breaks locally and ive really changed nothing.
Am I misunderstanding that chlorine/chloramine poisoning isnt a quick process? IMO keeping anything alive over 10yrs is a feat in itself with regular things like power outtages and equipment failure and i have plenty heads here over the decade mark living in what is most likely chlorine/chloramine source water being doused right on them 24/7.
Ultimately, what is the exact kind of test i need to be doing in my tanks to see if im dealing with chlorine or chloramine and if its even worth mitigating.
Im just not seeing anything like loss of appetite or stunted growth, cloudy eyes, labored breathing etc…
A good rule of thumb to tell if your fishes have been poisoned by chlorine is if they are "gasping for air" at the top of your tank/water line with no visible sign nor history of illness & no new additions to the tank right after a water change. Usually for smaller fish (<4") this is a 90% death sentence but medium->larger fish can typically still recover if you notice this fast enough within the hour. This is because a fish's breath isn't a vaccum where the water is pulled over the gills 24/7 every second but on a cycle + chlorine won't be uniformly present nor in every breath they take.
If you want to know if your fish(es) have had chronic long-term exposure (several days) the same behavior would occur but just on a longer timescale with the fishes "breathing" more "heavy" and becoming more lethargic instead of gasping at the top of the waterline if the concentration was low. Of course this is also a general indicator of onset illness so a water change + proper dechlorination should happen regardless. However, if it was a fairly high but not deadly level of chlorine, they would survive long enough for their slime coat production to go into overdrive causing it to sloughing off of themselves as the skin gets burned from the chlorine. At that point your chances of saving the fish is probably a 30/70 odd where you have to immediately put them in a chlorine clean tank, push a massive amount of air in the water, and pray that any infectious micro-organism is not present in the aquarium.
From what you've posted so far nothing about your fish scream any type of chlorine poisoning.
In terms of testing you can only really use OTC strips or liquid tests. If you've got expendable income you could get a spectrophotometer like this: https://hannainst.com/free-and-total-chlorine-ultra-high-range-portable-photometer-hi97771.html but it's a bit much IMO for most hobbyist. While it is the most accurate form of testing compared to all other available tests you need to constantly buy new reagents + ensure you're cleaning the glass containers extremely thoroughly to prevent any residual residues from sticking around unless you're okay with inaccurate tests.
IMO stressing about dechlorination is a lost cause. Not only is it an easy process, most common solutions (Seachem Prime & sodium thiosulfate) have a safety factor of at least 5x. So long as you're not building an automatic contraption that holds enough dechlorinator to surpass that safety factor you should be fine. Even then, as previously mentioned, the worst case scenario is just dissolved oxygen depleteion so if you have a running airpump/stone it's really hard to cause death with an overdose.
If I were in your exact scenario where I couldn't trust local water reports but had no desire to spend hundreds upfront I'd just buy test strips, fill a cup with water from the source i'd use before hand, and then test with 3 strips. I'd logged each test value (guessing based on color), average them out, and then treat based on the the volume of water I'm replacing with a 50g buffer (i.e. if I were treating 200g with liquid prime I'd use 25mL total, 20mL for the 200g replaced and an extra 5mL as safety). With a historic log set i'd only worry if the average during the water change was significantly off from previous readings or if there is a major trend shift. Of course with more expensive tests I'd only perform it once but the main thing is creating and logging a history if the local water report can't be trusted.
My exact water change method is the following:
1) Turn off pumps + heaters and prep water hose and water-removal tubing.
2) Drain water then remove the equipment
3) Pre-dump all measured dechlorinator in the area of the tank the tap water will be coming in
- I don't even measure chlorine and just assume 4ppm since I have a ton of sodium thiosulfate left over but a safe bet is assuming at least 1-2ppm of chlorine from your water line if you live in the US and are connected to a large city's waterline. I do this because 4 ppm is the legal EPA limit of chlorine in the US but YMMV if you live outside of it. If you're using Seachem Prime the dosage is based on 1-2ppm so just follow the label + add a buffer.
- I do pre-mix my sodium thiosulfate though in a small 1 cup container of tank water; stirring until no salt crystals are present.
- I do highly suggest users to get their local municipitality water report to check how much chlorine/chloramine to treat for unless you have specific reasons not to do so and understand what risks you take.
5) Turn on pumps immediately + wait 5-10mins then the heaters.
In my 20 years of fish keeping I have never lost a fish due to chlorine after doing these exact steps with Seachem safe (both liquid and powder) and sodium thiosulfate. In my earlier years I even forgot a couple of times to pre-dump the dechlorinator and still experienced no fish loss although I would suggest against it now if possible. Not because there is a huge risk but rather there is no reason to wait and possibly risk it. The only way to make this anymore idiot-proof is to pre-mix the dechlorinator with your water source.
Now you may be wondering why is chlorine poisoning not present in my fish based on all my previous posts. Simple, as mentioned before chlorine poisoning is a concentration AND probability problem. Your fish will only display symptoms of chlorine poisoning if a significant amount of the molecules reach your fish and actually pass through their cell walls resulting in organ damage. Additionally, when you're injecting your tank with untreated tapwater not all of the expected chlorine will be distributed at once. In other words, as the water pressure causes the new and old water to mix the chlorine will immediately hit water saturated with dehlorinator in addition to the flow spreading those dechlorinating molecules out to the rest of the tank. This results in an near 0% chance for any significant concentration of free chlorine to accumilate significant damage to any of my fish. Of course I recognize that not pre-mixing my water source with dechlorinator does mean that some free chlorine could likely reach my fish I also understand that the effect it has on them is effectively the same as scratching my arm because of an itch. Sure some cells may die but they will get replaced with no significant damage to the body.
Of course I would only suggest doing this on a well-established tank otherwise your bacteria colonies could die off since I immediately turn on my water pumps to get everything flowing again to reduce the chance of DO depletion.
HTH
Thanks again
T
thiswasgone

 … really just curious at this point. Free/Total chlorine test OTW…
… really just curious at this point. Free/Total chlorine test OTW…
Not stressing about using dechlor, just trying to see if its worth the investment for an inline chemical injector and the dechlor itself. Not terrible around $300-350 for the injector and a years worth of safe powder but thats a good chunk of change up front. Im not doing regular/physical water changes is my thing tho… EVER. I think thats y we may b comparing apples to oranges.
If for some reason i need extra water for maintenance or “emergency” i pull it out of another tank and let the autodrip fill that system back up. I can pull 50-400gallons out of several systems while there still running and they fill back up as they normally would over 24-48hrs etc.
Fully understand dumping hundreds of gallons directly into a tank in an hour is going to need to be chemically treated. Im dripping 3-4 gallons over an hour into 650 gal for ex.…If i can detect chloramine/chlorine in the tank i can justify the money. If not, i dont see the point in treating anything. Its like scratching ur arm like u said.
Not stressing about using dechlor, just trying to see if its worth the investment for an inline chemical injector and the dechlor itself. Not terrible around $300-350 for the injector and a years worth of safe powder but thats a good chunk of change up front. Im not doing regular/physical water changes is my thing tho… EVER. I think thats y we may b comparing apples to oranges.
If for some reason i need extra water for maintenance or “emergency” i pull it out of another tank and let the autodrip fill that system back up. I can pull 50-400gallons out of several systems while there still running and they fill back up as they normally would over 24-48hrs etc.
Fully understand dumping hundreds of gallons directly into a tank in an hour is going to need to be chemically treated. Im dripping 3-4 gallons over an hour into 650 gal for ex.…If i can detect chloramine/chlorine in the tank i can justify the money. If not, i dont see the point in treating anything. Its like scratching ur arm like u said.
Just to be clear
the term Total Chlorine is a term some water facilities use for Chloramine
the term free chlorine is used for simple chlorine, the use of chlorine gas, or sodium hypochlorite., etc, etc.
Sometimes you will see the terms both used because for instance (at the water plant where is was chemist)
Free chlorine at the water intake to prevent zebra and quagga mussels from building up.
But Total Chlorine (aka Chloramine) was injected after coagulation, sedimentation, filtration to provide disinfection throughout the entire distribution system
the term Total Chlorine is a term some water facilities use for Chloramine
the term free chlorine is used for simple chlorine, the use of chlorine gas, or sodium hypochlorite., etc, etc.
Sometimes you will see the terms both used because for instance (at the water plant where is was chemist)
Free chlorine at the water intake to prevent zebra and quagga mussels from building up.
But Total Chlorine (aka Chloramine) was injected after coagulation, sedimentation, filtration to provide disinfection throughout the entire distribution system
10/4 … Thank youJust to be clear
the term Total Chlorine is a term some water facilities use for Chloramine
the term free chlorine is used for simple chlorine, the use of chlorine gas, or sodium hypochlorite., etc, etc.
Sometimes you will see the terms both used because for instance (at the water plant where is was chemist)
Free chlorine at the water intake to prevent zebra and quagga mussels from building up.
But Total Chlorine (aka Chloramine) was injected after coagulation, sedimentation, filtration to provide disinfection throughout the entire distribution system
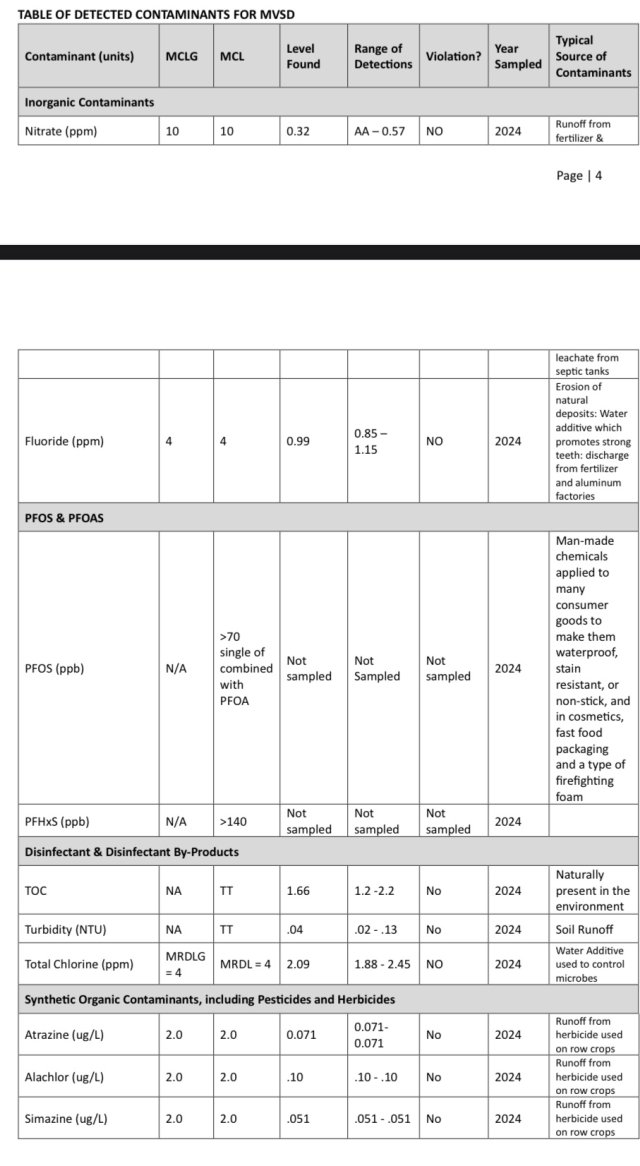
Water report i posted above shows the “total chlorine” twice at 2.06-2.09 ppm so that can actually mean chloramine. Got it lol…
Thoroughly surprised now because that would mean ive been dripping chloramine water for at least 2 years now in pretty heavy amounts. Im assuming even longer. Pretty lucky i guess as ive had no issues with fish sized .5”-43”…
The minimal amount of fresh water added slowly has to be my saving grace
if you have enough organic material, in the system (filter, sump, tank etc), that beneficial bacteria detoxifies the ammonia part of the ion, and the chlorine part of the ion is also oxidized by the organics.
So yes the slow drip, into a large amount of tank water could be the ticket.
And if you drip into sump, that will add extra protection.
So yes the slow drip, into a large amount of tank water could be the ticket.
And if you drip into sump, that will add extra protection.
Happy im not entirely crazy… as always, id rather be lucky than goodif you have enough organic material, in the system (filter, sump, tank etc), that beneficial bacteria detoxifies the ammonia part of the ion, and the chlorine part of the ion is also oxidized by the organics.
So yes the slow drip, into a large amount of tank water could be the ticket.
And if you drip into sump, that will add extra protection.
Thank you