- Have you tested your water?
- Yes
- If yes, what is your ammonia?
- 0
- If yes, what is your nitrite?
- 0
- If yes, what is your nitrate?
- 0
- If I did not test my water...
- ...I recognize that I will likely be asked to do a test, and that water tests are critical for solving freshwater health problems.
- Do you do water changes?
- Yes
- What percentage of water do you change?
- 0-10%
- How frequently do you change your water?
- Every week
- If I do not change my water...
- ...I recognize that I will likely be recommended to do a water change, and water changes are critical for preventing future freshwater health problems.
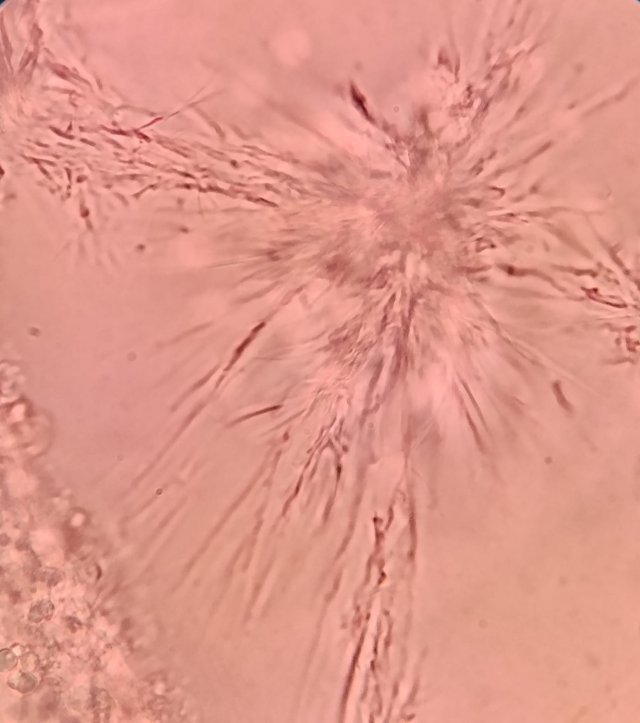
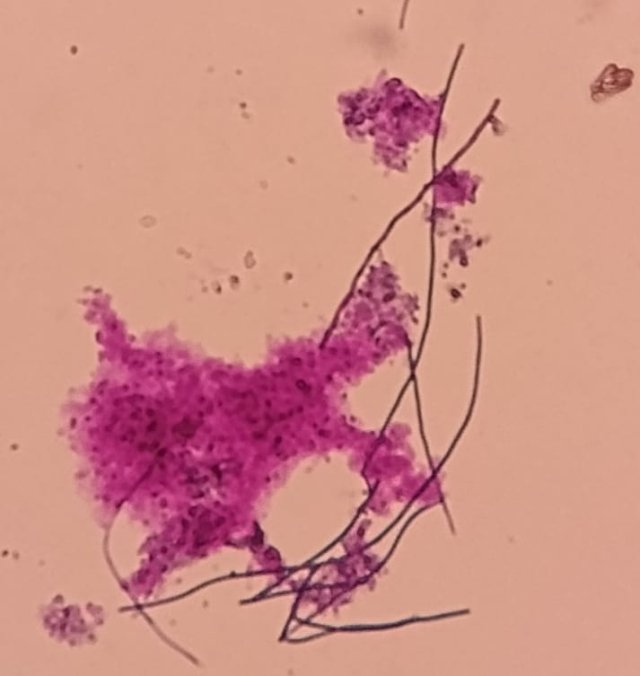
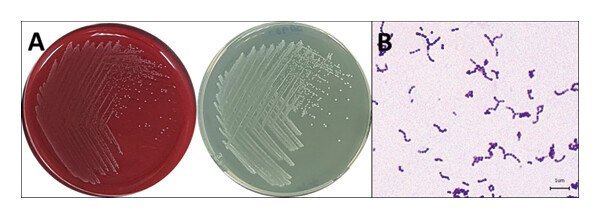
Dear MFK lovers; Please, could you identify these fungus? I am wondering if the second image are fungus hyphae or maybe celulose fibers, please help me! Both images are 100x, the first image looks like a spore burst where primary hyphae appear, but I don't know if it could also be an effect of the sample drying on the slide. Thank you!